The short answer? Absolutely not. Non-opioid topical relief solutions are experiencing unprecedented growth, innovation, and clinical validation in 2025. Despite skeptics questioning their long-term viability, emerging clinical data and policy frameworks demonstrate that topical therapies have never been more relevant to modern healthcare.
For healthcare providers managing active patients and professionals seeking evidence-based alternatives, the landscape has shifted dramatically. Let’s examine what the latest research reveals about the future of topical relief solutions.
The Clinical Renaissance of Topical Therapies
Healthcare providers are witnessing a fundamental transformation in how topical relief solutions are developed, tested, and implemented. The trajectory for non-opioid topical solutions points toward continued growth and refinement, with ongoing research focused on optimizing delivery systems and therapeutic effectiveness.
Recent clinical developments have established topical therapies as legitimate first-line interventions rather than supplementary treatments. Capsaicin and lidocaine patches now represent established non-opioid medications for managing subacute and chronic conditions, supported by robust clinical evidence and widespread provider adoption.
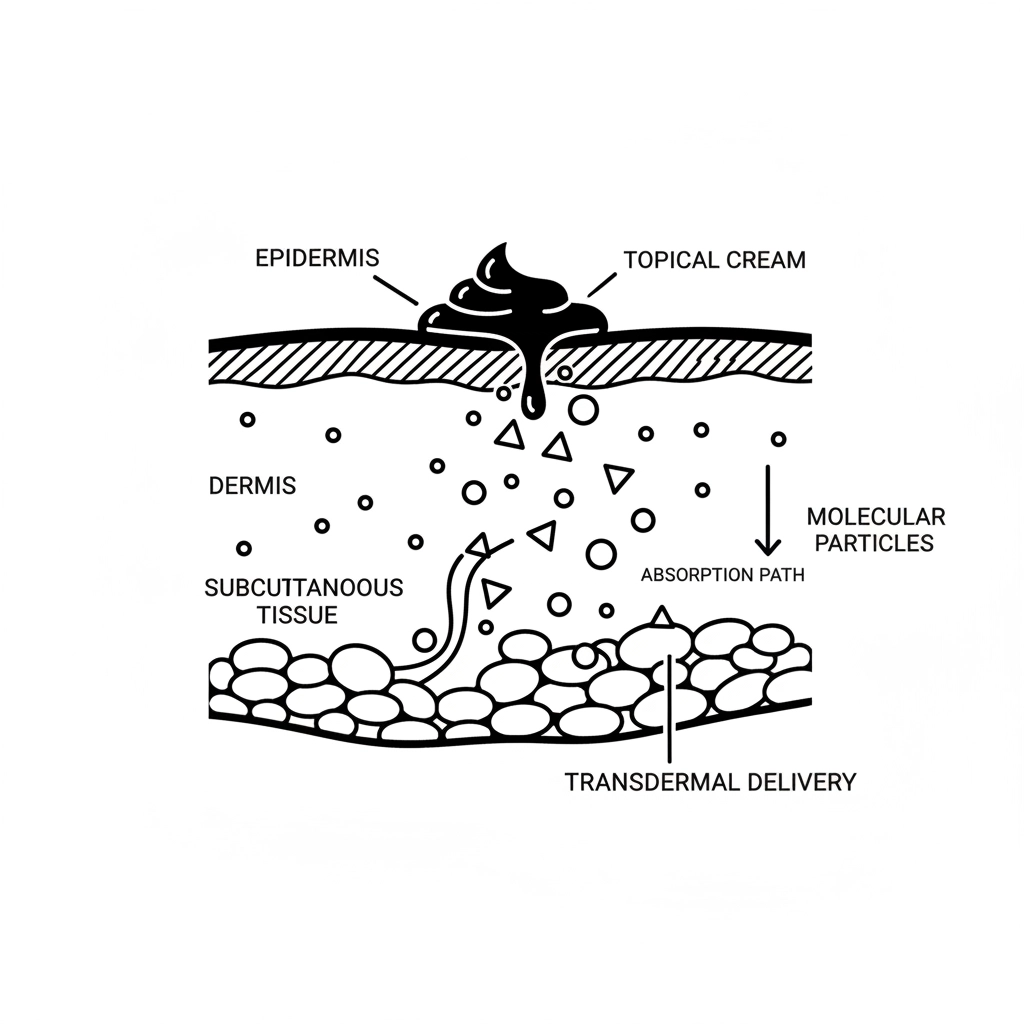
This isn’t merely about providing temporary cooling sensations: modern topical solutions target specific neurological pathways to address underlying mechanisms of action. Advanced formulations leverage transdermal delivery systems that ensure consistent, localized therapeutic concentrations while minimizing systemic exposure.
Policy Framework Validation: The NO PAIN Act Impact
The legislative landscape has fundamentally endorsed topical relief solutions through the NO PAIN Act, which explicitly includes topical therapies such as capsaicin and lidocaine patches among reimbursable treatment options. This policy-level recognition represents more than administrative convenience: it signals institutional confidence in topical modalities as clinically necessary interventions.
Healthcare administrators and providers can now confidently integrate topical solutions into treatment protocols, knowing that reimbursement frameworks support their use. This removes a significant barrier that previously limited adoption among cost-conscious practices and health systems.
For active adults and professionals, this means greater access to targeted relief options without the systemic risks associated with oral medications. The policy framework acknowledges what clinical practitioners have observed: topical solutions deliver measurable outcomes with favorable safety profiles.
Revolutionary Delivery Technologies Driving Growth
The 2025 clinical landscape showcases remarkable advances in topical delivery mechanisms that directly address historical limitations of absorption and bioavailability. Modern formulations utilize nanotechnology, liposomal encapsulation, and enhanced permeation techniques to optimize therapeutic delivery.

These technological innovations solve fundamental challenges that previously limited topical effectiveness:
Enhanced Bioavailability: Advanced delivery systems ensure therapeutic compounds reach target tissues in clinically relevant concentrations, eliminating concerns about surface-level application without meaningful penetration.
Sustained Release Profiles: Modern formulations provide controlled, extended therapeutic delivery, maintaining effective relief over hours rather than minutes.
Targeted Mechanism Activation: Rather than generic numbing effects, contemporary topical solutions can selectively modulate specific receptor pathways, including endocannabinoid system interactions and neuroprotective mechanisms.
Reduced Systemic Exposure: Localized action minimizes systemic absorption, reducing potential interactions and side effects commonly associated with oral alternatives.
Clinical Data Highlights: What 2025 Research Reveals
Recent clinical studies demonstrate significant therapeutic advantages for topical relief solutions across multiple application areas. Healthcare providers report improved patient satisfaction, reduced need for systemic medications, and enhanced functional outcomes when integrating modern topical therapies.
Key findings from 2025 clinical research include:
- Faster Onset Times: Advanced delivery systems achieve therapeutic effects within 15-30 minutes of application, comparable to oral alternatives
- Improved Compliance: Patients demonstrate higher adherence rates with topical solutions compared to multi-dose oral regimens
- Reduced Healthcare Utilization: Practices implementing comprehensive topical protocols report decreased follow-up appointments and enhanced patient self-management
- Enhanced Quality of Life Metrics: Active adults using evidence-based topical solutions show improved mobility scores and activity participation rates

These outcomes aren’t theoretical: they represent measurable improvements documented through standardized clinical assessment tools and patient-reported outcome measures.
Healthcare Provider Implementation Strategies
Successful integration of non-opioid topical relief solutions requires systematic approach that considers patient selection, application protocols, and ongoing monitoring. Healthcare providers should establish clear guidelines that maximize therapeutic benefits while ensuring safety and compliance.
Patient Assessment Framework:
- Evaluate localized areas requiring targeted intervention
- Review current medication regimens to identify potential systemic burden reduction opportunities
- Assess patient mobility goals and activity requirements
- Document baseline functional status for outcome tracking
Application Protocol Development:
- Establish standardized application techniques ensuring optimal coverage and penetration
- Provide clear patient education regarding proper use and expectations
- Implement documentation systems tracking therapeutic response and functional improvements
- Schedule appropriate follow-up intervals for efficacy assessment and protocol adjustment
Quality Assurance Considerations:
- Source products from manufacturers with transparent lab testing and quality verification
- Verify THC-free formulations when utilizing hemp-derived compounds
- Maintain current knowledge of FDA regulatory updates affecting topical therapeutics
- Document clinical outcomes supporting continued use and potential protocol expansion
The Active Adult Population: A Growing Market Reality
Active professionals, weekend athletes, and health-conscious individuals represent a significant population seeking effective relief solutions that don’t compromise their lifestyle or performance goals. Traditional oral alternatives often present concerns about cognitive effects, gastrointestinal impact, or activity restrictions that make topical solutions increasingly attractive.
This demographic values:
- Rapid Onset Without Systemic Effects: Ability to maintain mental clarity and physical coordination while addressing localized areas of concern
- Flexible Application Timing: Freedom to apply targeted relief before, during, or after activities without scheduling restrictions
- Professional Compatibility: Solutions that don’t interfere with work performance or require activity modifications
- Long-term Safety Profile: Confidence in sustained use without accumulating systemic burden or dependency concerns
Healthcare providers serving active populations find that modern topical solutions offer competitive therapeutic advantages while aligning with patient lifestyle preferences and safety expectations.
Innovation Pipeline: What’s Next for Topical Therapeutics
The clinical development pipeline for non-opioid topical solutions includes several breakthrough technologies that will further solidify their position in modern healthcare. Research focuses on precision delivery mechanisms, expanded therapeutic targets, and personalized formulation approaches.
Emerging developments include:
- Smart Delivery Systems: Formulations that respond to physiological conditions to optimize therapeutic release
- Combination Therapies: Multi-mechanism approaches targeting different aspects of the relief pathway simultaneously
- Personalized Formulations: Customized therapeutic profiles based on individual patient characteristics and response patterns
- Enhanced Biomarker Integration: Real-time monitoring capabilities that allow providers to track therapeutic effectiveness and adjust protocols accordingly
The Verdict: Thriving, Not Surviving
Non-opioid topical relief solutions have transitioned from alternative options to mainstream therapeutic modalities supported by clinical evidence, policy frameworks, and technological innovation. Healthcare providers and active adults alike benefit from expanded access to targeted, effective interventions that address relief needs without systemic compromises.
The question isn’t whether topical solutions will survive: it’s how quickly healthcare systems can adapt to incorporate these evidence-based tools into comprehensive care strategies. For practices focused on providing patients with science-backed, effective relief options, the current landscape offers unprecedented opportunities to deliver superior outcomes through modern topical therapeutics.
The future of relief isn’t just topical; it’s transformational. Explore advanced topical formulations that represent the cutting edge of non-opioid therapeutic innovation, backed by rigorous science and designed for real-world clinical success.