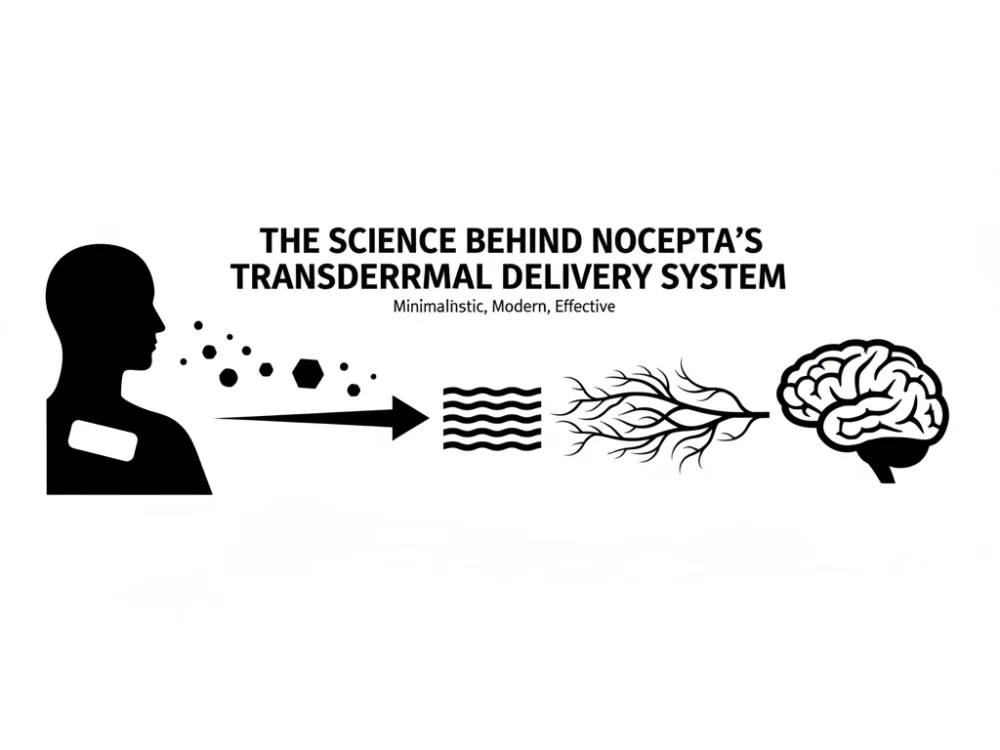
The landscape of discomfort management has evolved dramatically over the past decade. With increasing scrutiny of opioid-based approaches, healthcare professionals and patients alike have been seeking alternatives that provide effective relief while minimizing systemic risks. This shift has sparked an important question: Are non-opioid topical solutions still relevant, or have they been overshadowed by newer therapeutic approaches?
The answer might surprise you. Far from being obsolete, non-opioid topical solutions are experiencing a renaissance, backed by expanding clinical evidence, regulatory support, and growing healthcare provider confidence.
The Current State of Non-Opioid Topical Solutions
Recent clinical practice guidelines demonstrate unwavering support for topical approaches in modern discomfort management. The CDC’s 2022 Clinical Practice Guideline specifically recognizes that non-opioid therapies are at least as effective as opioids for many common types of acute discomfort conditions. This isn’t just theoretical support: it represents a fundamental shift in how healthcare providers approach patient care.
Topical solutions, including capsaicin and lidocaine formulations, maintain their position as recommended interventions for both subacute and chronic discomfort conditions. These recommendations aren’t based on convenience alone; they reflect accumulated clinical evidence supporting the efficacy and safety profile of targeted topical applications.
The mechanism of action for advanced topical formulations involves localized delivery of active compounds directly to affected tissues. This targeted approach allows for concentrated relief at the application site while minimizing systemic exposure: a critical advantage in today’s healthcare environment where patient safety and risk mitigation remain paramount concerns.
Legislative Support: The 2025 NOPAIN Act
Perhaps the strongest indicator of continued confidence in non-opioid topical solutions comes from recent legislative developments. The 2025 NOPAIN Act represents a significant policy shift, specifically expanding reimbursement coverage for topical discomfort management options, including capsaicin and lidocaine patches.
This legislative support isn’t merely symbolic. Insurance coverage expansion reflects evidence-based recognition that topical solutions provide measurable patient benefits while reducing healthcare system costs associated with more invasive interventions. When healthcare payers: organizations intensely focused on cost-effectiveness: expand coverage for specific therapeutic approaches, it signals robust confidence in both clinical efficacy and economic value.
The Act’s provisions also acknowledge the role of topical solutions within comprehensive care plans, recognizing them as legitimate alternatives rather than supplementary options. This positioning elevates the clinical status of advanced topical formulations and validates their place in evidence-based practice protocols.
Clinical Integration and Provider Confidence

Healthcare providers increasingly incorporate topical solutions into integrated treatment approaches. Rather than relying on single-modality interventions, modern discomfort management emphasizes multi-modal strategies that may include topical applications alongside physical therapy, behavioral interventions, and other non-pharmacological approaches.
This integration reflects a sophisticated understanding of discomfort mechanisms and the recognition that targeted interventions can address specific aspects of the patient experience. Advanced topical formulations, particularly those utilizing transdermal delivery systems, offer healthcare providers a tool for addressing localized discomfort while supporting overall mobility and function.
The science behind modern topical solutions has evolved significantly beyond simple cooling gels or basic analgesic preparations. Contemporary formulations leverage nanoemulsion technology, enhanced permeation systems, and precise delivery mechanisms that optimize active ingredient penetration and bioavailability at target sites.
Patient Trust and Adoption Patterns
Patient acceptance of topical solutions remains strong, driven by several key factors:
Perceived Safety Profile: Patients appreciate the localized application approach, which aligns with growing preferences for targeted rather than systemic interventions. The ability to apply treatment directly to areas of discomfort provides patients with a sense of control over their care.
Convenience and Accessibility: Topical applications integrate seamlessly into daily routines without the scheduling constraints or lifestyle modifications required by other therapeutic approaches. This accessibility factor contributes significantly to patient compliance and long-term adoption.
Transparent Ingredient Profiles: Modern consumers increasingly seek transparency in their healthcare products. Advanced topical formulations that provide clear ingredient information and explain mechanisms of action build trust through education and transparency.
Innovation in Topical Delivery Systems
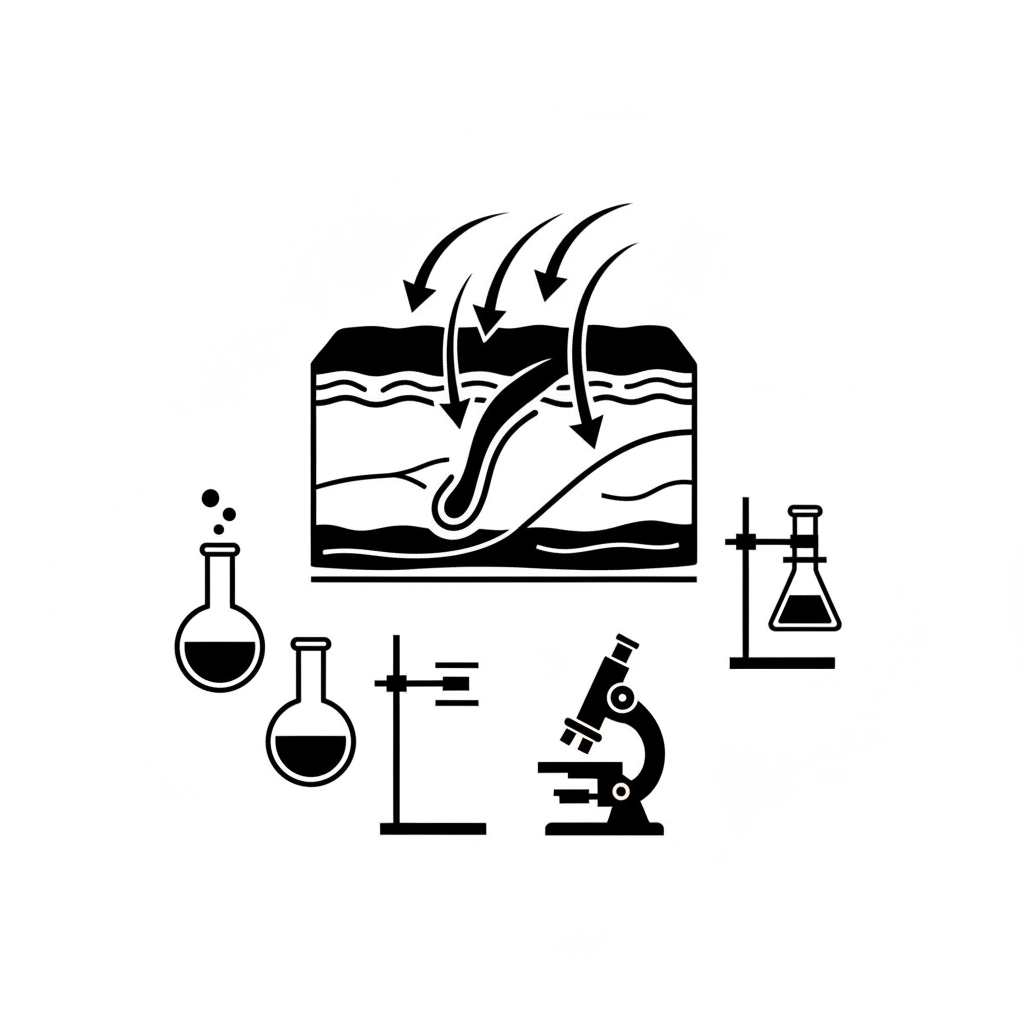
The topical solutions sector continues to experience significant innovation, particularly in delivery system technology. Transdermal delivery systems now utilize sophisticated approaches including:
Nanoemulsion Technology: These systems create ultra-small particle sizes that enhance skin permeation and improve bioavailability of active compounds. This technology allows for more effective delivery of beneficial ingredients to target tissues.
Enhanced Permeation Systems: Advanced formulations incorporate penetration enhancers that optimize the movement of active ingredients through skin barriers, improving efficacy while maintaining safety profiles.
Sustained Release Mechanisms: Modern topical solutions can provide extended relief through controlled-release formulations that maintain therapeutic levels over extended periods, reducing application frequency and improving patient convenience.
These technological advances address historical limitations of topical applications, such as limited penetration depth and short duration of action. Contemporary formulations provide clinically relevant relief while maintaining the safety advantages associated with topical delivery.
Evidence-Based Efficacy
Clinical research continues to validate the effectiveness of well-formulated topical solutions. Studies examining topical applications for various discomfort conditions consistently demonstrate:
Localized Action: Topical formulations can achieve therapeutic concentrations at application sites while minimizing systemic exposure, providing targeted relief where needed most.
Comparable Efficacy: Research indicates that appropriate topical interventions can provide relief comparable to oral medications for many localized discomfort conditions, often with fewer side effects.
Improved Function: Beyond symptom management, effective topical solutions support improved mobility and function, contributing to overall quality of life improvements.
Professional Integration Strategies

Healthcare professionals successfully integrate topical solutions into practice through several strategic approaches:
Comprehensive Assessment: Effective use begins with thorough evaluation of patient presentation, including location and characteristics of discomfort, previous treatment responses, and overall health status.
Patient Education: Successful outcomes require clear communication about proper application techniques, expected timelines for relief, and integration with other therapeutic interventions.
Monitoring and Adjustment: Professional oversight ensures optimal outcomes through regular assessment of response and adjustment of approach as needed.
Whether you’re a physical therapist, chiropractor, medical supplier, or other healthcare provider, incorporating evidence-based topical solutions into practice protocols can enhance patient outcomes while maintaining safety standards.
The Future of Topical Discomfort Management
The trajectory for non-opioid topical solutions points toward continued growth and refinement. Ongoing research focuses on optimizing delivery systems, identifying new active compounds, and developing formulations tailored to specific patient populations and discomfort patterns.
Regulatory agencies continue to recognize the value of topical approaches, with streamlined approval pathways for innovative formulations that demonstrate safety and efficacy. This regulatory support encourages continued innovation while maintaining appropriate oversight.
Healthcare economics also favor topical solutions, as they typically offer cost-effective relief compared to more intensive interventions, supporting their continued integration into value-based care models.
Conclusion: A Thriving Future
Far from being obsolete, non-opioid topical solutions represent a thriving sector of discomfort management with strong clinical support, expanding coverage, and continued innovation. The combination of legislative backing, clinical evidence, and technological advancement positions these solutions as cornerstone interventions in modern healthcare.
The trust placed in topical solutions by healthcare providers, patients, and payers reflects their demonstrated value in supporting comfort and mobility while maintaining favorable safety profiles. As the healthcare landscape continues to evolve toward personalized, evidence-based care, topical solutions stand ready to meet the challenge.
For healthcare professionals seeking to provide patients with science-backed, targeted relief options, advanced topical formulations offer a proven pathway to improved outcomes. Explore the power of modern transdermal delivery systems and discover how targeted topical interventions can enhance your practice’s ability to support patient comfort and mobility.